The compliance gap in medical AI
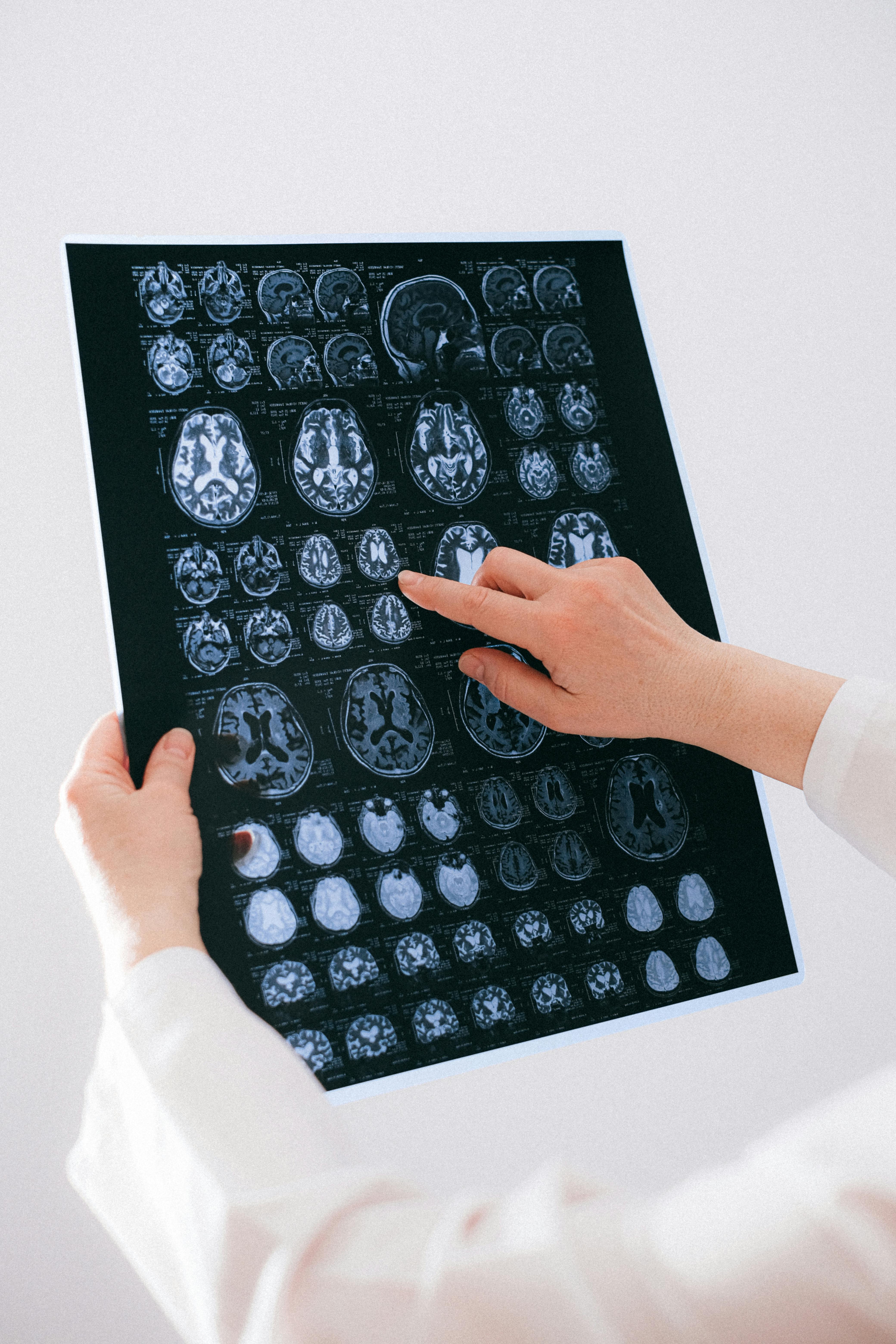
AI is increasingly responsible for building medical devices — writing diagnostic algorithms, generating training data, even drafting regulatory submissions. But the tools for verifying this software haven't kept pace.
Traditional QA processes were designed for deterministic software. They can't evaluate whether a training dataset is representative, whether a model is robust against adversarial inputs, or whether AI-generated code meets IEC 62304 safety classifications.
That's the gap CarexData fills. We bring AI-native compliance verification to the teams building the next generation of medical technology.

Built on research, designed for industry
CarexData was founded by a team with PhDs in Computer Science and deep experience in medical imaging AI research. Our compliance methodology bridges the gap between academic rigor and regulatory reality.
The CarexData Compliance Score (CCS) evaluates AI medical software across six dimensions — fidelity, representativeness, robustness, documentation, security, and transparency — with each dimension directly traceable to specific regulatory requirements.
Our methodology is open and peer-reviewable. We believe the standard for medical AI compliance should be as transparent and evidence-based as the devices it evaluates.